News
Can Artificial Intelligence save the NHS?
April 2023
In recent months, it has been almost impossible to escape reports of the staffing crisis in the NHS and the impact this is having on the services the NHS can provide.
At the same time, it seems that questions about what OpenAI’s ChatGPT can be used for have permeated almost every discussion forum.
At the very least, it’s clear that there is huge potential for new and diverse applications of AI.
Such discussions have led us to wonder whether AI can play a significant role in automating general patient diagnosis within a clinical setting to directly alleviate the pressure on front line staffing, and further whether EP patent law in this area is having any impact.
Can using AI to triage patients, triage the NHS itself?
This is clearly a complex and highly charged question.
However, it’s clear that for AI-based general diagnosis to even be considered a possibility, the reliability of an AI diagnosis must reach a level at least rivalling that of human medical professionals (notwithstanding whether patients will ever be comfortable with integrating AI into common place clinical services).
At the same time, a take-away message from the move to remote care necessitated by the Covid pandemic was the importance of conducting a holistic assessment of a patient.
In order for an AI-based system to be able to usefully diagnose a range of conditions in patients, we might then consider a holistic assessment of a patient to be a key requirement.
However, the quality of any AI’s output is governed by how well the AI is trained and training an AI to perform a holistic assessment of a patient not only requires truly vast amounts of data, but also an incredibly thorough understanding of biases and limitations within the training data set.
How can researchers be motivated to invest the resources required to achieve this level of training?
This is where patents can come into play, but how, and are they doing enough?
The underlying principle of the patent system is the basic transaction of a monopoly right in exchange for a public disclosure of an inventive contribution to the relevant field. A proponent of the patent system will argue that this monopoly right provides the financial motivation for businesses and even lone inventors to outlay the significant investments in time and resources that drive innovation.
While it is possible for software applications, such as AI, to be patentable, assuming they meet the requirements for novelty and inventive step (for example, providing a real-world effect, or improving how a computer-system runs), for our particular example case of an AI for performing medical diagnosis, additional restrictions to patentability apply.
Under patent law in the UK and Europe, the granting of the monopoly right of a patent to motivate investment is considered to be outweighed by the need for medical professionals to be able to perform their duties. Thus, specific restrictions apply that prohibit the patenting of ‘methods of diagnosis’. As a result, a patent claim to an AI explicitly performing a method of diagnosis is not generally allowable.
1This is an over-simplification of the requirements that must be met for software to be patentable. However, the exclusions of ‘method of diagnosis’ are the most relevant for this article.
While there are some ways around this exception that may enable patent protection to be sought for inventive contributions of AI that aid in the diagnosis of a patient, for example, claims directed to the AI itself as a tool, rather than the full diagnostic method performed when the AI is used as the tool, there is a risk when filing an application in this technology area that the protection of the AI may be limited.
Thus, in Europe, the patent protection for an AI used for a method of diagnosis is not as complete as it may be for AI applications in other fields, for example, AI used for e.g., image recognition in self-driving vehicles.
In different jurisdictions, such as the US, the law attempts to address the problem of balancing motivation to innovate with protection of medical professionals in a different way.
Under US patent law, a method of diagnosis may be patented, but no action may be taken against medical professionals to enforce such a patent.
It may not be unreasonable to assume then that the limitations in European law may not be maximising the incentive for companies that primarily operate in Europe to invest in this area of AI.
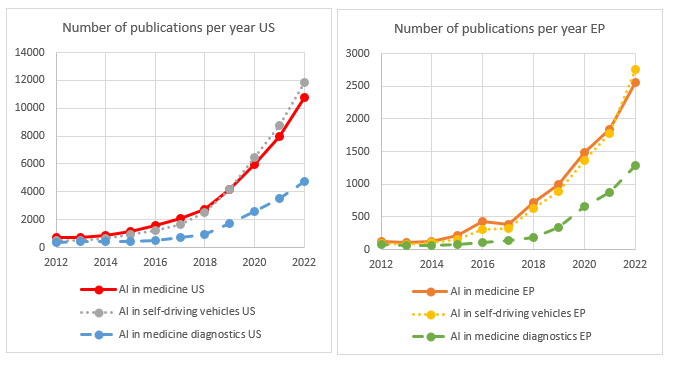
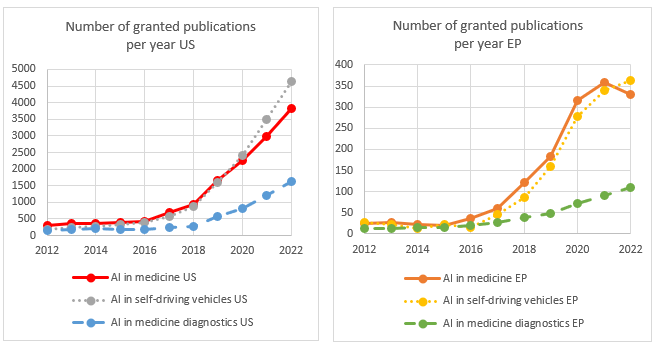
We have analysed the number of European versus US patent filings published within the last 10 years using keyword searching to identify patent filings involving the use of AI for medical applications in general (and medical diagnosis in particular) and compared these results to patents identified as using AI for self-driving vehicle applications, which is used here as a baseline for a technology area where AI is also readily adopted, but where the additional patenting restrictions for diagnosing a patient do not apply.
Interestingly, our assumption about the consequences of the limitations in European law doesn’t appear to be the case.
2Credit to Gleb Kolpakov, Data Scientist, HGF.
In fact, from the above, in comparison with the US, where the number of published and granted patents applying AI in the medical sector appears to have fallen behind patent filings applying AI to self-driving vehicles in recent years, in Europe, the patent filings published and granted in these two technology areas are keeping pace with each other.
Thus, it doesn’t appear that inventors or businesses that primarily operate in Europe have been any less motivated to apply for patents using AI for general medical uses or medical diagnosis, compared to their US counter-parts.
However, the evidence may suggest that in Europe, the success rate of granted patents for AI applied specifically to medical diagnosis may be lagging behind the US.
As a result, perhaps this instead highlights the value a patent firm like HGF, who are highly experienced in navigating the exclusions in European law may add when drafting and prosecuting patents in this field. If you would like to discuss this topic in more detail, or any other medical device patent advice needs, please feel free to contact either of the authors – Partner, Jeff Clarke, and Associate, Victoria Bruce.
This article was written by Partner Jeff Clarke and Associate Victoria Bruce for HealthTech World. Read the original article here.






























